
Strictly speaking, recombinant DNA refers to DNA molecules, while molecular cloning refers to the experimental methods used to assemble them. 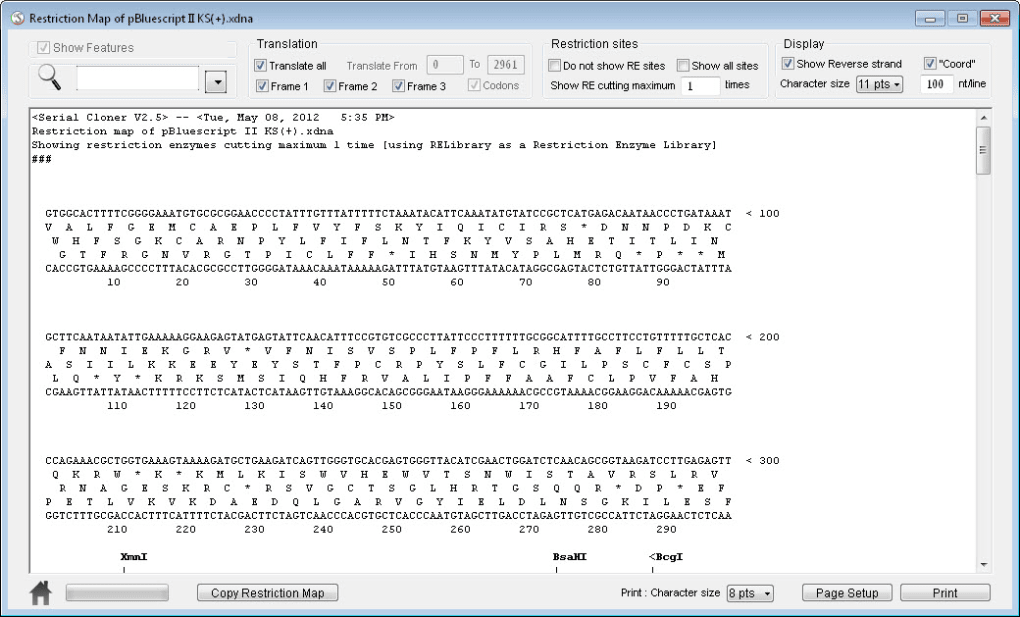
Thus, both the resulting bacterial population, and the recombinant DNA molecule, are commonly referred to as "clones". This single cell can then be expanded exponentially to generate a large amount of bacteria, each of which contain copies of the original recombinant molecule. This process takes advantage of the fact that a single bacterial cell can be induced to take up and replicate a single recombinant DNA molecule. Because they contain foreign DNA fragments, these are transgenic or genetically modified microorganisms (GMO). This will generate a population of organisms in which recombinant DNA molecules are replicated along with the host DNA. The recombinant DNA is then introduced into a host organism (typically an easy-to-grow, benign, laboratory strain of E. Subsequently, these fragments are then combined with vector DNA to generate recombinant DNA molecules. In a conventional molecular cloning experiment, the DNA to be cloned is obtained from an organism of interest, then treated with enzymes in the test tube to generate smaller DNA fragments. Molecular cloning methods are central to many contemporary areas of modern biology and medicine. Molecular cloning generally uses DNA sequences from two different organisms: the species that is the source of the DNA to be cloned, and the species that will serve as the living host for replication of the recombinant DNA. The use of the word cloning refers to the fact that the method involves the replication of one molecule to produce a population of cells with identical DNA molecules. Molecular cloning is a set of experimental methods in molecular biology that are used to assemble recombinant DNA molecules and to direct their replication within host organisms. Either Genbank-formatted file can most likely be used for conversions in the future and this is it is probably best to just keep the latest.Diagram of molecular cloning using bacteria and plasmids. This is probably best as the UGENE project will have a Genbank-formatted file in the project folder and it could become confusing. It is advised that you save it in a manner to clearly distinguishable from the SERIAL CLONER files, optionally even in a separate folder.įor my approach, I use File > Export Project, give it a good name, and save the project in a new directory.Īt this point, you can delete the Genbank-formatted file generated by Serial Cloner. I prefer to save each sequence I work on as a separate UGENE project, but you can choose whatever approach works for you work style. Save the file so you can open it in UGENE later without needing to repeat the conversion. Step 2: Save as UGENE project or document format and clean up gb extension but you may not see this depending on your system's view settings for extenstions.) Under File > Export > Export As GenBank and save, preferably with a distinguishing name. xdna to the Genbank format.įind your Serial Cloner file ending in. If you haven't done so already, you'll need to convert from the Serial Cloner file format that ends in extension. Step 1: locate the Serial Cloner file and generate the Genbank files you'll need Main thing is you need a GenBank-formatted file generated from Serial Cloner. The process of going from Serial Cloner to UGENE seems much simpler as the feature annotations in UGENE display by default. The annotated features from Serial Cloner will ultimately be intact and displayed in UGENE if you follow this process. This describes how to take a Serial Cloner sequence with annotated features and move it into Unipro UGENE with your favorite text editor and minimal effort. Converting a Serial Cloner sequence to Unipro UGENE
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
